HTA Council Recommendation on Bivalent COVID-19 Vaccines as booster Date of OIC-SOH Approval: 25 April 2023 This assessment followed the HTA Council evaluation framework to evaluate COVID-19 vaccines using the following criteria: (1) responsiveness to magnitude and severity; (2) clinical efficacy and safety; (3) affordability, viability and feasibility; (4) household financial impact; (5) social impact; and continue reading : HTA Council Recommendation on Bivalent COVID-19 Vaccines as Booster
[Announcement] HTA Council Releases Its Preliminary Recommendation on the Financing of DOACs for Nonvalvular Atrial Fibrillation
The Health Technology Assessment (HTA) Council releases its preliminary recommendation on the government financing of apixaban, dabigatran, and rivaroxaban for the prevention of myocardial infarction, cerebrovascular diseases and other cardiovascular events among patients with nonvalvular atrial fibrillation (NVAF) and subgroups of NVAF patients through its inclusion in the Philippine National Formulary.Appeals will be accepted from continue reading : [Announcement] HTA Council Releases Its Preliminary Recommendation on the Financing of DOACs for Nonvalvular Atrial Fibrillation
[Announcement] HTA Council Releases Its Preliminary Recommendation on the Non-Financing of Cerebrolysin for Post Ischemic Stroke
The Health Technology Assessment (HTA) Council releases its preliminary recommendation on the non-government financing of cerebrolysin in combination with rehabilitation or standard of care for the treatment of adults post-ischemic stroke through its non-inclusion in the Philippine National Formulary.Appeals will be accepted from 17 March 2023 to 31 March 2023 Kindly see full advisory:CALL FOR continue reading : [Announcement] HTA Council Releases Its Preliminary Recommendation on the Non-Financing of Cerebrolysin for Post Ischemic Stroke
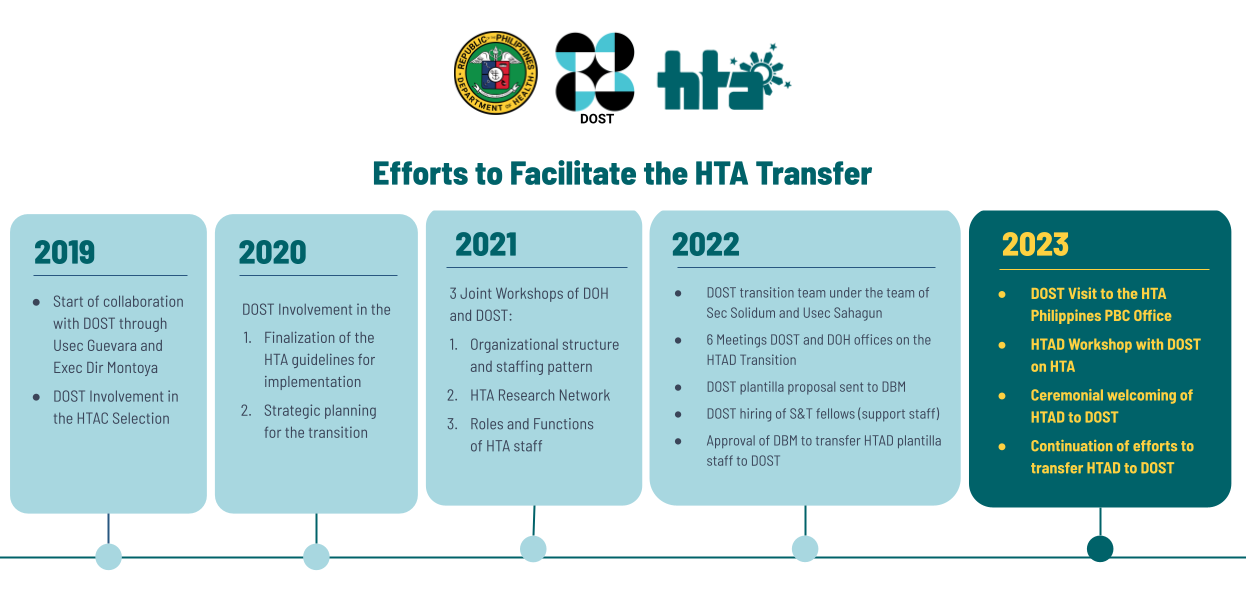
HTA Philippines Guidance: Efforts to Facilitate the HTA Transfer
The Department of Health (DOH) officially transferred the operations of the Health Technology Assessment (HTA) Philippines to the Department of Science and Technology (DOST) through a ceremonial turnover of the HTA Council and HTA Division from the DOH to the DOST on 06 March 2023 at the Philippine International Convention Center (PICC), Manila. To facilitate continue reading : HTA Philippines Guidance: Efforts to Facilitate the HTA Transfer
HTAC Guidance: PNF Inclusion of Recombinant Human Erythropoietin (Epoetin Alfa) 4000 IU/mL Solution for Injection (IV/SC) 1mL Pre-filled Syringe
(Updated as of 14 March 2023) On 24 January 2023, the OIC-Secretary of Health (SOH) approved the Health Technology Assessment Council (HTAC) recommendation for the government financing of recombinant Human Erythropoietin (epoetin alfa) 4000 IU/mL solution for injection (IV/SC), 1mL pre-filled syringe, for the management of chemotherapy-induced anemia in patients with non-myeloid disease through its minor inclusion continue reading : HTAC Guidance: PNF Inclusion of Recombinant Human Erythropoietin (Epoetin Alfa) 4000 IU/mL Solution for Injection (IV/SC) 1mL Pre-filled Syringe
